17 Apr, 2026 | ronald | No Comments
The Joe Tippens Protocol: What It Is and Why MillionsAre Searching for It
In 2016, an Oklahoma businessman named Joe Tippens was given three months to live. By 2017, scans showed no detectable cancer in his body. His account of taking a cheap dog-deworming drug — fenbendazole — alongside conventional treatment sparked a global movement that has since touched millions of lives, filled pharmacy shelves with shortages, and launched an entirely new field of cancer research.
Whether you are a patient, a caregiver, a researcher, or simply someone who stumbled across this story on social media, you deserve a thorough, honest account of what happened, what the science says, and what questions still need answering. This article is that account.
The Story That Started Everything
In August 2016, a businessman from Oklahoma, Joe Tippens, was diagnosed with small-cell lung cancer and underwent a clinical trial under the supervision of his oncologist. He was informed of a short life expectancy — from three months to one year.
A veterinarian recommended he try fenbendazole along with vitamin E supplements, cannabidiol (CBD) oil, and bioavailable curcumin — all while continuing the clinical trial. A positron emission tomography (PET) scan after three months did not detect any cancer cells anywhere in his body.
Tippens shared his story in 2017 through a personal blog called MyCancerStory.rocks and through YouTube videos. The response was extraordinary. Within a few years, his account had been read, watched, and shared millions of times — not just in the United States, but in South Korea, Japan, India, and across Europe.
Tippens claimed fenbendazole helped him achieve remission from stage IV small cell lung cancer when combined with curcumin and CBD-enriched cannabis oil. However, Tippens’ narrative often omits a critical detail: he was also enrolled in a clinical trial for Keytruda — a PD-L1 checkpoint inhibitor immunotherapy known to produce complete responses in some patients.
This context matters enormously — and we’ll return to it. But first, let’s understand exactly what the protocol involves.
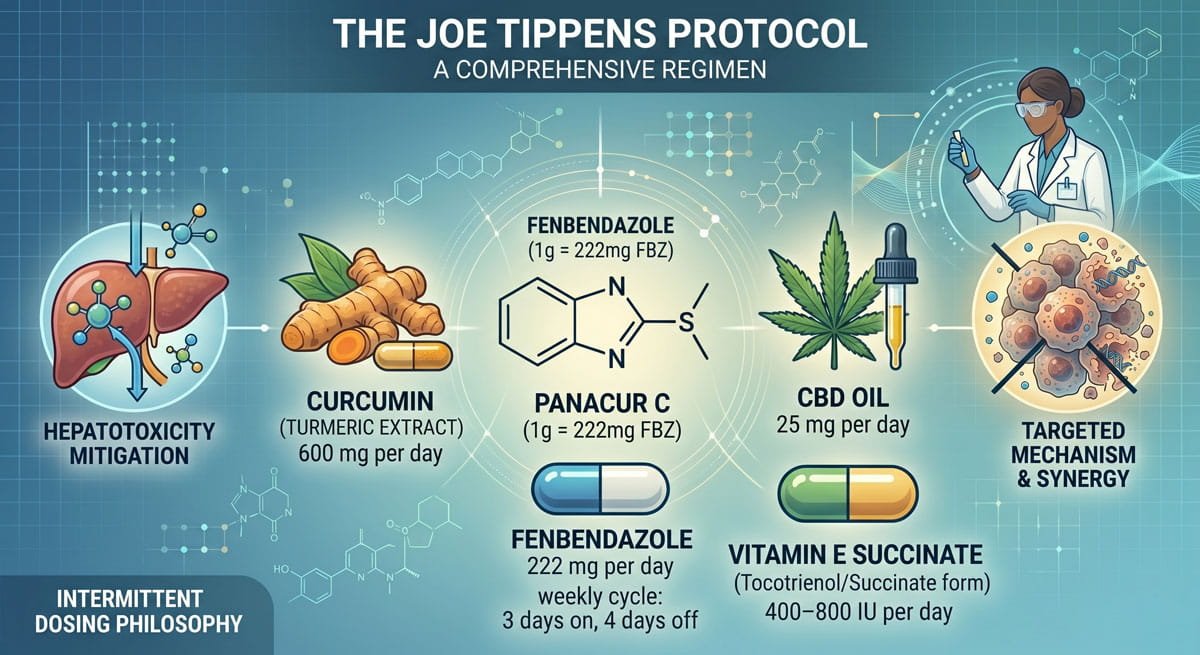
What Is the Joe Tippens Protocol?
The original fenbendazole protocol was pioneered by Joe Tippens after his terminal small cell lung cancer diagnosis. As documented in a 2022 PLoS ONE retrospective study, it involves taking 222 mg of fenbendazole per day for 3 consecutive days, followed by 4 days off. The regimen also typically includes 600 mg of curcumin and 25 mg of CBD oil daily.
The Original Joe Tippens Protocol
The core philosophy of this protocol was intermittent dosing. By cycling the medication — 3 days on, 4 days off — the objective was to maintain therapeutic efficacy while mitigating potential hepatotoxicity, as fenbendazole is metabolized extensively by the liver.
How Has the Protocol Evolved?
Joe Tippens first shared his story in 2017. Over time, the protocol’s composition and presentation have changed as new supplements were added and online communities formed around it. None of these versions have undergone formal clinical testing. Several online versions now include combinations of vitamin E, curcumin, CBD oil, and berberine — consumer-created additions, not verified updates by medical professionals.
In a pivotal 2025 case series published in Case Reports in Oncology, researchers documented patients achieving sustained remission by taking 222 mg daily — without the 4-day rest period — and without reported adverse effects. A patient with Stage IV prostate cancer utilized a dose range of 222–444 mg/day over 26 months, achieving near-complete remission alongside androgen deprivation therapy.
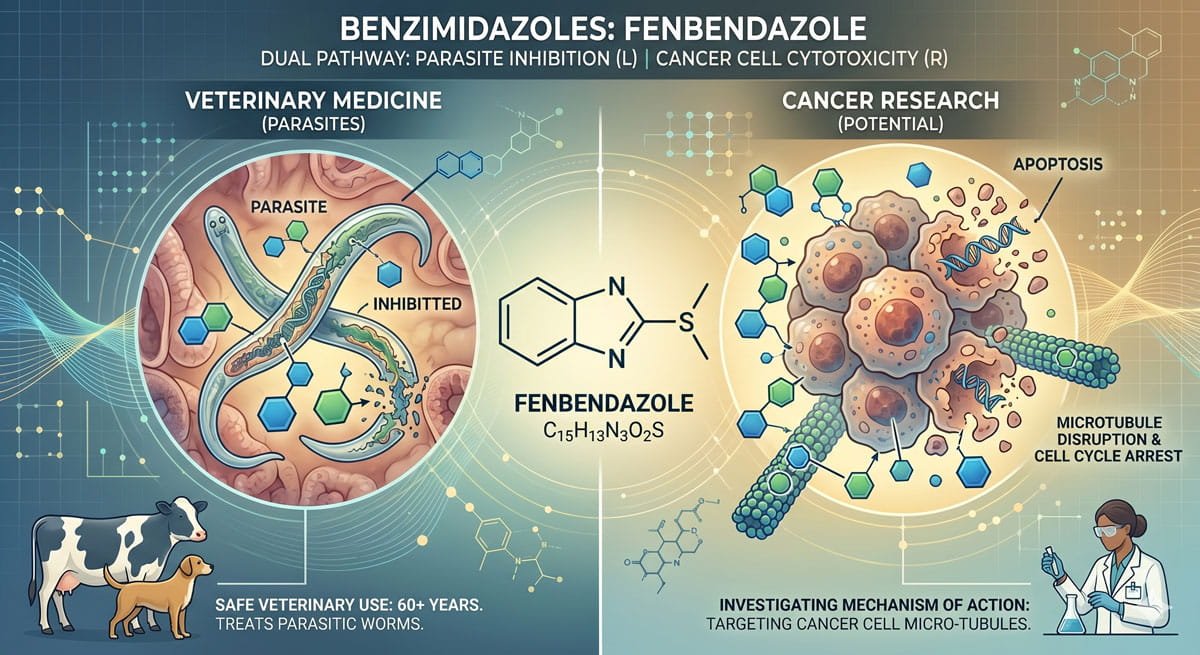
What Does the Science Actually Say?
Fenbendazole belongs to a drug family called benzimidazoles — compounds that have been used safely in veterinary medicine for over six decades. Researchers have been studying whether its mechanism of action in parasites might also apply to cancer cells.
FBZ binds to β-tubulin, disrupting microtubule formation in dividing cells. This is the same mechanism used by several established chemotherapy drugs like taxanes and vinca alkaloids.
Cancer cells rely heavily on glucose (the Warburg effect). Studies show FBZ may inhibit glycolysis pathways in tumor cells, starving them of energy.
Multiple cell-line studies demonstrate FBZ triggering programmed cancer cell death — including G2/M cell cycle arrest — in colorectal, lung, breast, and cervical cancer models.
Research suggests FBZ may stabilize the p53 tumor suppressor protein, which plays a critical role in preventing abnormal cell proliferation.
Early lab studies show fenbendazole suppresses growth in tumor cells from cancers such as breast, prostate, colorectal, lung, and liver cancer. Any future cancer therapy use will require clinical trials, blood tests, and liver function monitoring to confirm therapeutic efficacy and patient safety.
By inhibiting glycolysis in cancer cells and preventing lactate buildup, fenbendazole surpasses albendazole and mebendazole in treating drug-resistant cells, making it the benzimidazole of choice for cancer research. Despite numerous success stories and the extensive research performed in vitro and in vivo, repurposing fenbendazole for cancer treatment remains non-suggested by conventional medical institutions and oncologists.
How One Story Spread Across the Globe?
The journey from one man’s blog post to a worldwide phenomenon is itself a remarkable story of the internet age.
The Unanswered Question: Was It Really the Fenbendazole?
This is the most important question — and the one most online accounts skip past. The honest answer is: we don’t know.
Tippens’ outcome may have been driven by genetic factors such as microsatellite instability or a high mutational burden, which are strongly associated with favorable responses to immunotherapy. For instance, a study published in the New England Journal of Medicine reported a 100% complete response rate in similar cases treated with checkpoint inhibitors.
These personal “success” stories can sound compelling, but they do not prove fenbendazole is a safe and effective cancer treatment. It is impossible to know whether each person’s outcome was due to fenbendazole or something else, including the other treatments they were taking for their cancer.
“It’s impossible to know whether the outcome was due to fenbendazole or something else, including other treatments the person was taking.”— American Cancer Society, What to Know About Fenbendazole (2025)
Laboratory studies of fenbendazole and other anthelmintic drugs have shown some early promise against a variety of cancers. But treatment that works against cancer cells in a petri dish or in animals doesn’t always work in people. The results from human studies are still very early and mixed. Much more research is needed to answer key questions about fenbendazole.
Known Risks and Safety Considerations
Fenbendazole has a generally favorable safety profile in animals. But using it in humans — especially alongside chemotherapy or immunotherapy — carries real risks that any responsible discussion must include.
Key Reported Risks: Liver enzyme elevation and drug-induced liver injury have been documented in case reports of people using fenbendazole. Fenbendazole may interact with chemotherapy or immunotherapy agents. Poor water solubility means orally administered FBZ may not reach sufficient systemic levels to impact tumors. Product quality varies widely counterfeit and adulterated products have been reported in the market.
Fenbendazole’s Cancer research is preliminary, and most reports involve self-administration outside clinical supervision. It is not FDA-approved for cancer treatment. Self-medication risks include drug-induced liver injury and other adverse effects, especially with uncontrolled dosing or concurrent supplements. Dose variability in human use is unstandardized; monitoring with blood tests and liver function panels is essential in research settings.
Where Does the Research Stand in 2025–2026?
The scientific conversation around fenbendazole has moved considerably — but formal clinical trials in humans are still largely absent, which is the core gap researchers are calling for urgently.
A review of 170 anecdotal reports from 2023–2025 noted tumor regression and remission across various cancers, though these lacked clinical rigor. Social media platforms, including Facebook groups with over 100,000 members, amplify these claims, driving off-label use.
Clinical trials should be funded and performed to promote the possible application of fenbendazole as an inexpensive, well-characterized, and widely available anticancer therapeutic in animals and humans.
To advance fenbendazole research, scientists have proposed Phase I trials initiating dose-escalation studies in patients with advanced, treatment-refractory cancers to establish safety and pharmacokinetics, followed by Phase II randomized trials in high-priority cancers such as lung and ovarian to assess efficacy. Combination studies evaluating FBZ with immunotherapies like PD-1 inhibitors are also a priority.
Frequently Asked Questions
Is Joe Tippens still alive in 2026?
As of January 2026, Joe Tippens was reported to be alive and in remission from cancer. In February 2026, he attended the Annie Appleseed Conference in West Palm Beach.
What exactly is fenbendazole?
Fenbendazole is a benzimidazole anthelmintic commonly used to treat a variety of animal parasitic infections. It is an inexpensive antiparasitic drug widely used in veterinary medicine, readily accessible through animal supply stores and online platforms. The patent expired in the early 1990s, making it available as a generic drug.
Did the fenbendazole cure Joe Tippens’ cancer?
This cannot be determined. Tippens was simultaneously enrolled in a clinical trial for Keytruda (pembrolizumab), a powerful immunotherapy drug. Medical experts note his recovery may be attributable to the immunotherapy, genetic factors, or a combination of multiple factors — not fenbendazole alone. No controlled trial has isolated fenbendazole’s individual contribution.
What does the original protocol include?
Joe Tippens used the following treatment regimen: curcumin 600 mg per day, cannabidiol oil 25 mg per day, and fenbendazole 222 mg per day for 3 consecutive days with four-day intervals in between.
Is fenbendazole FDA-approved for human cancer treatment?
No. As of 2026, fenbendazole remains a veterinary medication with no FDA approval for human use in any indication, including cancer. Any use by humans is off-label and should be discussed with a licensed physician.
Where can I buy pharmaceutical-grade fenbendazole?
Look for products that provide a Certificate of Analysis (CoA), clearly state purity levels (99%+), are manufactured under pharmaceutical-grade standards, and come from a reputable, transparent supplier. BuyFenbendazole.us offers lab-verified fenbendazole with full documentation.
Explore Pharmaceutical-Grade Fenbendazole
Every batch at BuyFenbendazole.us is independently lab-tested for 99%+ purity. Certificate of Analysis included. Fast US shipping with same-day fulfillment.
Shop Now at BuyFenbendazole.us →
For research and informational purposes only. Not FDA-approved for human use. Consult your physician.

Write Reviews
Leave a Comment
No Comments & Reviews